Sf4 Dipole Moment - How To Discuss
By John Johnson •
Sf4 Dipole Moment
Is there a dupole moment in SF4? 3
Is there a dupole moment in SF4?
Are all bonds the same and consist of polar bonds?
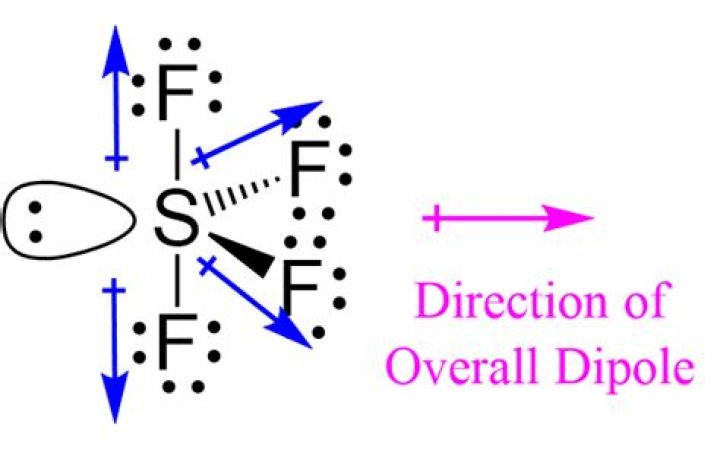
SF4 has a dupole moment because it has not only covalent bonds but also a pair of electrons. The electrons of a molecule have a partial negative charge at the end of the pair, and a partial positive charge at the end of the bond of the molecule, so yes, it is usually polar.
All connections are identical and polar. The electronegativity difference between S and F is not very large, but the bond is still slightly polar.
Sf4 dupole moment
Sf4 Dipole Moment
Sf4 Dipole Moment
This page can help you.
D:
Is there a dupole moment in SF4?
Is there a dupole moment in SF4?
Are all bonds the same and consist of polar bonds?

