Nicl2 - How To Discuss
By William Burgess •
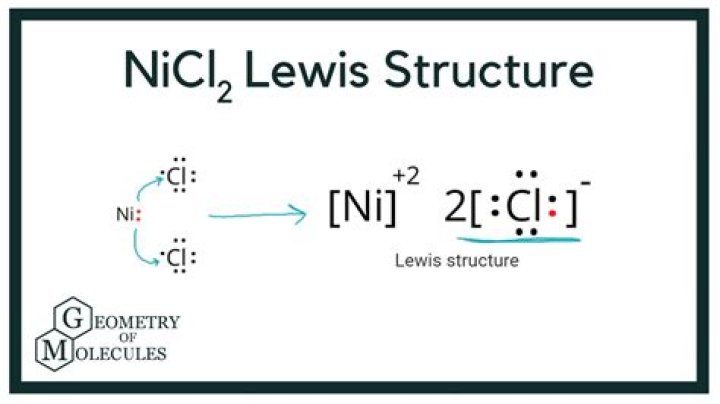
Nicl2
Need help! Acidic, basic or neutral NiCl2?
NiCl2 (s) + 6H2O Â Â [Ni (H2O) 6] 2 + + 2Cl (blue)
C (aq) is not significantly hydrolyzed (HCl vs. strong acid)
[Ni (H2O) 6] 2 + + H2O Â Â [Ni (H2O) 5 (OH)] + + [H3O] +
pKa ~ 10 makes it slightly acidic
Nicl2
Nicl2
Need help! Is NiCl2 Acidic, Basic or Neutral? 3
I have no idea. Wikipedia says it is an acid, so what is the hydrolysis reaction?
Thank you very much!
NiCl2 (s) + 6H2O Â [Ni (H2O) 6] 2 + + 2Cl (blue)
Cl (aq) is not significantly hydrolyzed (HCl vs. strong acid)
[Ni (H2O) 6] 2 + + H2O Â [Ni (H2O) 5 (OH)] + + [H3O] +
pKa ~ 10 makes it slightly acidic.

