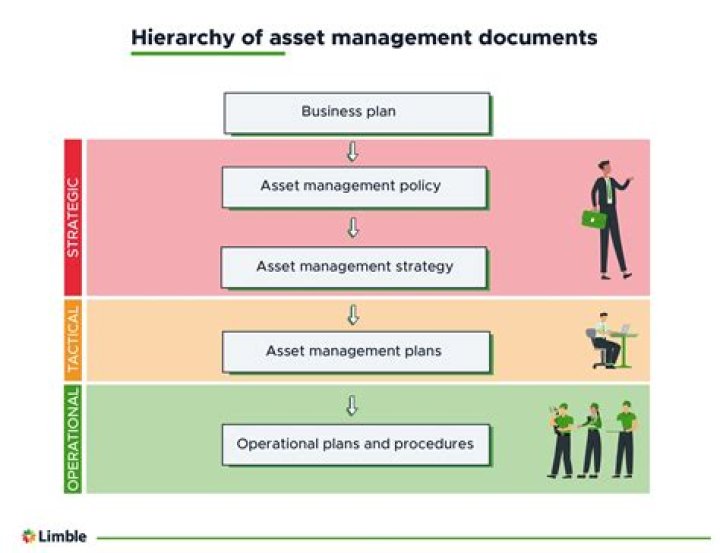
Nickel Valence Electrons - How To Discuss
Nickel Valence Electrons
How many valence electrons are there in nickel? 3
I bet 3, then 4 and my answer is wrong. I'm so stupid
update1, 2, 3 and 4 all tried wrong.
Nickel Valence Electron
Nickel Valence Electrons
Nickel Valence Electrons
Nickel Valence Electron
This page can help you.
D:
How many valence electrons are there in nickel?
I bet 3, then 4 and my answer is wrong. I'm so stupid
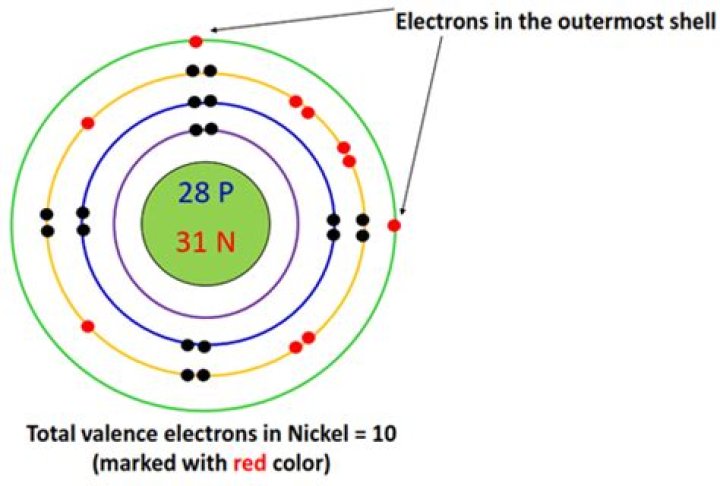
Nickel has 10 valence electrons. Neutral atoms have two sequences of electrons with the same energy: [Ar] 3dà ¸4s² and [Ar] 3dà ¹4s¹ In both cases, all ten electrons are from the valence electron of Noble gas, [Air] Are out. Many of your interviewers consider the concept of oxidation state in terms of the number of valence electrons, which are two different things.
At the center of the matrix ic, for example, from Sc # 21 to Zn # 30, new electrons go to the ad in INNER S. They do not add new valence electrons. So the answer is 2 for all of them, just like for # 20.
Ni has 28 electrons because its outermost s has 2 electrons, it has 2 valence electrons.
Ni (28) = 1s2.2s2.2p6.3s2.3p6.3d8.4s2
Nickel is a transition element.
So there are 2 electrons outside that can join the compound.
Therefore, nickel is traditionally called 2. valence electron.
Then
Nickel usually has an oxidation number of +2 in compounds.
But one of the 3D electrons is sometimes more in the moving nickel atom.
Therefore, the oxidation state of nickel can be +3.
If someone gives you a good answer.
Consider a better answer.
This is the only award we have received.
The correct answer is ten (!). Ni [Ar] 3d 8 4s 2. In Ni (CO) 4 (Google Nickel Carbonyl), for example, all the electrons are stored in bonds, either through bonds or.
Nickel Valence Electrons
Nickel Valence Electrons
Ni (28) = 1s2.2s2.2p6.3s2.3p6.3d8.4s2
This means that it has 2 valence electrons [2 verbal 3s electrons]
It has 10 limit electrons! In Table ic only the column number is calculated for this intermediate area. So in Sc 3, in Ti 4, in V 5 and so on. So if you count ... nothing (nickel) is 10! What helps, for example.