Lattice Energy Of Cabr2 - How To Discuss
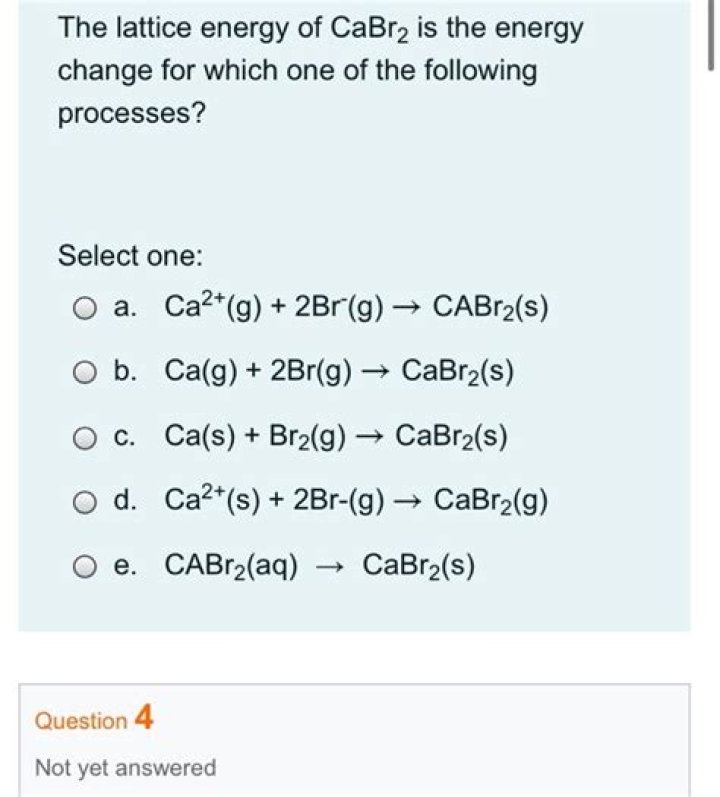
Lattice Energy Of Cabr2
Calculate grid energy? Someone explain! 3
1. Use the following thermodynamic data to calculate the grid energy of CaBr2 (s).
Trim Value (kJ / mol)
Delta {H} f [CaBr2 (s)] 675
Delta {H} f [Ca (g)] 179
I1 (Ca) 590.
I2 (Ca) 1145
E (Fr) 325
Hayes' law is a reference. That is, the change in energy is independent of this path. (Using D instead of Delta)
So Df (CaBr2) = Detomization (Ca) + I1 (Ca) + I2 (Ca) + 2 X Detomization (Br2) + 2 X EA (Br) + Tissue Energy (CaBr2)
Therefore, 675 = + 179 + 590 + 1145 + (2 x 112) + (2 x 325) + L.E. (CaBr2)
Then i go (CaBr2) = 6751795901145224 + 650 = 2163 kJ mol1
(PS: I also need bromine datum values)
Calculate network performance.
This page can help you.
D:
Calculate grid energy? Someone explain!
1. Use the following thermodynamic data to calculate the grid energy of CaBr2 (s).
Nominal term (kJ / mol)
Delta {H} f [CaBr2 (s)] 675
Delta {H} f [Ca (g)] 179
I1 (Ca) 590.
I2 (Ca) 1145
E (Fr) 325
Lattice Energy Of Cabr2
Lattice Energy Of Cabr2
Master the reaction chemistry.
2162 kJ / mol
Delta Hf * [CaBr2 (s)] 675
Delta-Hf * [Ca (g)] 179
Delta Hf * [Br (g)] 112
I1 (Ca) 590
I2 (Ca) 1145
E (Fr) 325
Answer: +2163 kJ / mol
Chemistry Master (Correct Answer; Sam is Correct)
Chemical reaction domain: 2163 kJ / mol
Lattice Energy Of Cabr2
Lattice Energy Of Cabr2
Sam answered well.
Cb has the correct number but no negative sign.
As Sam said, this is 2163 kJ / mol.
2193 kJ
Test 2163 kJ

