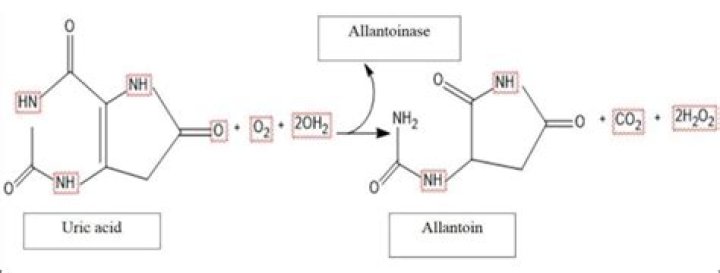
How to find limiting reagent
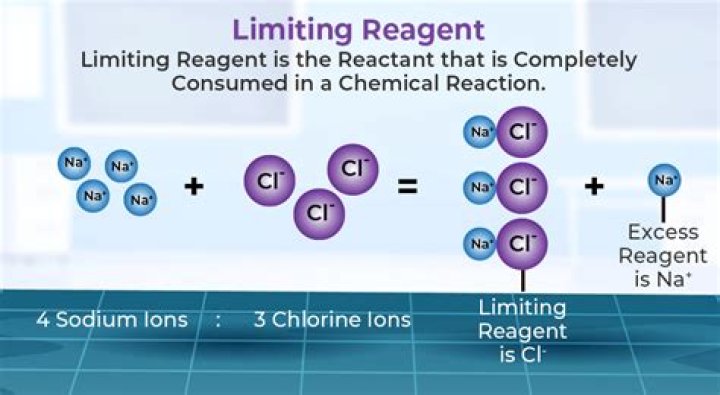
How do you identify a limiting reactant? An easy way to determine the limiting reactant is to calculate the number of ■■■■■ of reaction each will produce: divide the ■■■■■ of each reactant by its corresponding factor in a balanced equation. I did it for you in the table above. HCl is the limiting reagent because it gives the fewest number of reaction ■■■■■.
How do they know which is the limiting reagent?
One way to find a limiting reactant is to calculate the amount of product that can be obtained from each reactant. The reagent that produces the least amount of product is the limiting reagent. The following scenario illustrates the importance of limiting the amount of reagents.
Which of the reactants is the limiting reagent?
In a chemical reaction, a limiting reagent, also called a limiting reagent, is a substance that is completely consumed upon completion of the chemical reaction. The amount of resulting product is limited by this reagent because the reaction cannot proceed without it.
How will you identify a limiting reagent in a reaction?
How do you find the limiting reactant? First, find a balanced chemical equation for a particular chemical reaction. Then convert all the information to ■■■■■ (use the molar mass as the conversion factor). The next step is to calculate the molar ratio based on the information provided. Use the limit amount of reagent to calculate the amount of product produced.
How do you determine limiting reagent?
One way to determine the limiting reagent is to compare the molar ratios of the amounts of reagent used. This method is most useful when there are only two items. The limiting reagent can also be obtained by comparing the amount of product that can be generated from each reagent.
What is the formula for limiting reagent?
Calculate the limiting reactants. Use this limiting reagent calculator to calculate the limiting reagent for the reaction. To calculate the limiting reactant, enter the equation for the reactants and chemical reaction products and their coefficients will be displayed.
![:brown_circle: :brown_circle:]() How do you calculate excess reagent?
How do you calculate excess reagent?
To determine the excess of reactant, the first step is to calculate the ■■■■■ of each reactant in the reaction. The stoichiometry of the equation shows the relative number of ■■■■■ that would react in an ideal situation.
What is limited reactants?
Limiting Reagent. A limiting reagent (or limiting reagent) in a chemical reaction is a substance that is completely consumed upon completion of a chemical reaction. The amount of resulting product is limited by this reagent because the reaction cannot proceed without it.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How do you identify a limiting reactant in chemistry
How do you identify a limiting reactant in chemistry
The limiting reagent (or reagent) in the reaction is determined by calculating the amount of product produced by each reagent. The reagent that produces the least amount of product is the limiting reagent.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How do I find the limiting reactant?
How do I find the limiting reactant?
When you get the current number of ■■■■■ for each reactant and are asked to find the limiting reactant for a specific reaction, the easiest way to find the limiting reactant is to divide each value by the appropriate factor for that substance. balanced) the lower value is the limiting reagent.
How do you identify a limiting reactant definition
There are two methods to find a limiting reagent. The first is to compare the actual mole ratio of the reactants to the mole ratio of a balanced chemical equation. Another method is to calculate the mass of the product in grams obtained with each reagent. The reagent that produces the least mass of product is the limiting reagent.
How do you identify a limiting reactant in photosynthesis
Since there is an excess of oxygen, the amount of glucose is used to calculate the amount of product in the reaction. If more than 6 ■■■■■ of O2 per mole of C6H12O6 are available, there is an excess of oxygen and glucose is the limiting reagent. If less than 6 ■■■■■ of oxygen are available per mole of glucose, oxygen is the limiting reagent.
How to find the limiting reagent in a chemical equation?
When determining the limiting reagent, keep the following points in mind: If there are only two reagents, write a balanced chemical equation and check the amount of Reagent B needed to react with Reagent A. If the amount of reagent B is greater, reagent A is the limiting reagent.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which is a limiting factor in the process of photosynthesis?
Which is a limiting factor in the process of photosynthesis?
Photosynthesis is the process by which plants absorb sunlight and use it to convert carbon dioxide and water into glucose and oxygen. The main limiting factors in this process are light intensity, temperature and carbon dioxide content.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How does limiting reagent depend on mole ratio?
How does limiting reagent depend on mole ratio?
The limiting reagent depends on the molar ratio and not on the masses of the reactants present.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What's the difference between a reactant and a limiting reactant?
What's the difference between a reactant and a limiting reactant?
A reagent is a substance that directly enters a reaction and causes a reaction, and a reagent is a substance that participates in a chemical reaction. Why is the limiting reagent so important?
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How do you identify a limiting reactant worksheet
How do you identify a limiting reactant worksheet
To determine the limiting reactant, calculate the ■■■■■ of each reactant present and compare this ratio to the mole ratio of reactants in a balanced chemical equation.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the limiting reactant in the reaction shown?
What is the limiting reactant in the reaction shown?
A limiting reagent (or limiting reagent) of a chemical reaction is a substance that is completely consumed upon completion of a chemical reaction. This means that the reaction stops once the limiting reagent is completely consumed. As the name suggests, the limiting reagent limits the amount of product produced.
How does a limiting reactant affect a reaction?
The presence of a limiting reagent reduces the amount of products that a given reaction can form. A reagent that acts as a limiting reagent is consumed in the reaction first, essentially leaving the other reagent(s) behind. This means that the amount of product the reaction produces will depend on the limiting reagent.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How do you find a limiting reagent?
How do you find a limiting reagent?
Finding the Limiting Reactant: Procedure 1. Find the limiting reactant by looking at the ■■■■■ of each reactant. Find a balanced chemical equation for a chemical reaction. Convert all information to ■■■■■ (you will most likely use molar mass as the conversion factor).
What is a real life example of a limiting reactant?
Examples of limiting reagents. Note that there is 1 mole of oxygen and 1 mole of hydrogen that must go through the next reaction. Since the reaction uses hydrogen twice as fast as oxygen, hydrogen will be the limiting reagent. Example: 100 g zinc is mixed with 100 g hydrochloric acid. 14 Nov.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How do you identify a limiting reactant chart
How do you identify a limiting reactant chart
There are two methods to find a limiting reagent. The first is to compare the actual mole ratio of the reactants to the mole ratio of a balanced chemical equation. Another method is to calculate the mass of the product in grams obtained with each reagent.
![:brown_circle: :brown_circle:]() Why is the limiting reagent so important?
Why is the limiting reagent so important?
Wiki response. The limiting reagent/reagent is important because it can tell the chemist that with the amount of that substance he uses, because it limits the reaction, only x ■■■■■ of product can be formed instead of the theoretical y if the ideal amount is used.
Which compound would be the limiting reagent?
Benzene is then the limiting factor. This conclusion can be confirmed by comparing the molar ratio of O 2 and C 6 H 6 according to the balanced equation with the current current molar ratio: since the actual ratio is greater than required, O 2 is an excess of reactant, confirming that benzene is a limiting reagent.
![:brown_circle: :brown_circle:]() How do they know which is the limiting reagent definition
How do they know which is the limiting reagent definition
The reagent that is completely used in the reaction is called the limiting reagent. Limiting reagents are substances that are completely consumed when a chemical reaction is completed. They are also called limiting agents or limiting reagents.
How do they know which is the limiting reagent symbol
One way to find a limiting reactant is to calculate the amount of product that can be obtained from each reactant. The reagent that produces the least amount of product is the limiting reagent.
What is limiting reagent in chemistry?
Key points The limiting reagent is the fully used reagent. Given the balanced chemical equation describing the reaction, there are several ways to determine the limiting reactant. One way to determine the limiting reagent is to compare the molar ratios of the amounts of reagent used.
![:brown_circle: :brown_circle:]() How do you calculate excess reactants?
How do you calculate excess reactants?
The excess of reactants can be determined by a balanced chemical reaction equation which gives the molar ratio between the reactants. For example, if the balanced reaction equation is: 2 AgI + Na 2S → Ag 2S + 2 NaI.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() Which of the reactants is the limiting reagent definition
Which of the reactants is the limiting reagent definition
When the amount of reagent B is greater, reagent A is the limiting reagent. A reagent that is present in an amount less than the stoichiometric requirement is the limiting reagent. Another method of finding the limiting agent is to calculate the amount of product formed by each reactant.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() Which of the reactants is the limiting reagent formula
Which of the reactants is the limiting reagent formula
When the amount of reagent B is greater, reagent A is the limiting reagent. A reagent present in an amount less than the stoichiometrically required amount is a limiting reagent. Another method of finding a limiting agent is to calculate the amount of product produced by each reactant.
![:brown_circle: :brown_circle:]() What is the difference between a reactant and a reagent?
What is the difference between a reactant and a reagent?
Although the two terms have similar meanings, their roles are different in a given answer. The main difference between reagent and reagent is that reagents are compounds that are consumed and participate directly in a reaction whereas reagents are used to measure the magnitude of a chemical reaction or to observe a reaction.
How will you identify a limiting reagent in a reaction using
One way to determine the limiting reagent is to compare the molar ratios of the amounts of reagent used. This method is most useful when there are only two items. The limiting reagent can also be obtained by comparing the amount of product that can be generated from each reagent.
What exactly is a limited reagent?
Limiting Reagent. A limiting reagent (or limiting reagent or limiting agent) in a chemical reaction is a substance that is completely consumed upon completion of the chemical reaction. The amount of resulting product is limited by this reagent because the reaction cannot proceed without it.
![:brown_circle: :brown_circle:]() Theoretical yield
Theoretical yield
Theoretical yield: The maximum possible mass of a product that can cause a chemical reaction. It is calculated using molar ratios. Actual Yield: The mass of the product that causes the chemical reaction in real life. As a general rule, it is below the theoretical return for several reasons:
How do you calculate theoretical yield of something?
- Balance the chemical equation. Do you want to write to us?
- Express the mass of reactants in ■■■■■.
- Find the limiting reactant. The next step is to determine which of the two reactants is limiting.
- Find your theoretical return.
- Find your return rate.
![:brown_circle: :brown_circle:]() Why do you get theoretical yield less than actual?
Why do you get theoretical yield less than actual?
Usually this is below the theoretical performance. Causes: Incomplete reactions where some reactants fail to react to form a product. Is the actual return generally higher or lower than the theoretical? Theoretical results are expected results when each bit of each reagent reacts completely with the others. Actual profitability is lower than theoretical for several reasons. First, there is no 100% answer.
How do you calculate theoretical yield in chemistry?
Calculation of theoretical yield Determine the desired product. Write down the ■■■■■ of your limiting reagent. Find out the ratio of molecules in your product and reagent. Multiply this ratio by the number of reactants in ■■■■■. Convert the result to grams.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() How does actual yield and percent differ from theoretical yield?
How does actual yield and percent differ from theoretical yield?
Theoretical yield is the expected amount of product and the actual yield is the actual amount of product received in the lab. By calculating the percentage of recovery, you can determine how many experimental errors were made in the lab. Sometimes the actual profitability is higher than the theoretical.
![:brown_circle: :brown_circle:]() What is the benefit of having a limiting reagent?
What is the benefit of having a limiting reagent?
The limiting reagent for a chemical reaction is a substance that is completely consumed when the chemical reaction is completed. For example, the advantage would be that if you cancel the response in step c, the product can still be shipped. If you let it rise completely, it will crumble, which is not suitable for the application the dough is being used for.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is the concept of limiting reagent?
What is the concept of limiting reagent?
Limiting reagents are substances that are completely consumed when a chemical reaction is completed. They are also called limiting agents or limiting reagents. Depending on the stoichiometry of chemical reactions, a fixed amount of reagents is required to complete the reaction.
![:brown_circle: :brown_circle:]() Is there always a limiting reagent?
Is there always a limiting reagent?
The equations must not contain limiting reagents. The equations are purely theoretical expressions and are always expressed in ■■■■■. Restrictive reagents are found in real chemical reactions.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() How to find limiting reagent of reactant from balanced equation
How to find limiting reagent of reactant from balanced equation
The equations must not contain limiting reagents. The equations are purely theoretical expressions and are always expressed in ■■■■■. The equations are purely theoretical expressions and are always expressed in ■■■■■.
How do you determine the limiting reactant?
To determine which reactant is limiting, you must first determine how much product would be formed from each reactant if all the reactant were consumed. The reagent that produces the least amount of product is the limiting reagent.
![:brown_circle: :brown_circle:]() How to find percent yield
How to find percent yield
Interest formula. The equation for the percentage return is: percentage return = (actual return / theoretical return) x 100%. True: Actual yield is the amount of product produced by a chemical reaction.
![:eight_spoked_asterisk: :eight_spoked_asterisk:]() What is my percent yield and how is it calculated?
What is my percent yield and how is it calculated?
Interest income is the percentage of real income relative to fictitious income. It is calculated as the experimental yield divided by the theoretical yield multiplied by 100%. If the actual and theoretical returns are the same, the percentage return is 100%.
How does one calculate percentage yield?
Part 3 of 3: Calculate the interest rate. Read more about the yield percentage. The theoretical return you calculated assumes that everything went perfectly. Record the actual conduct of the experiment. If you did the experiment yourself, collect the purified product of your reaction and weigh it on a scale for calculation. Divide the actual yield by the theoretical yield.
![:brown_circle: :brown_circle:]() How can a percent yield be more than 100 percent?
How can a percent yield be more than 100 percent?
However, a yield of more than 100% is possible if the measured reaction product contains impurities that would make its mass greater than it would actually be if the product were pure. Whenever a chemist synthesizes a chemical of interest, he is always concerned with the purification of the reaction products.
![:brown_circle: :brown_circle:]() Which would be the limiting reagent in the reaction?
Which would be the limiting reagent in the reaction?
Therefore, hydrogen gas limits the reaction and is therefore considered as the limiting reagent for this reaction. Explanation of the limiting reagent. This reagent generally determines when the reaction stops. The exact amount of reagent needed to react with another element can be calculated from the stoichiometry of the reaction. The limiting reagent depends on the molar ratio and not on the masses of the reactants present.
![:brown_circle: :brown_circle:]() How to calculate theoretical yield
How to calculate theoretical yield
Theoretical yield is the mass of the product calculated using a balanced chemical equation. Percentage output = (actual output theoretical output) x 100. Optimal output is the best possible output obtained for the range of the given reaction conditions.
What is a good theoretical yield?
The ideal or theoretical efficiency of a chemical reaction will be 100%, an ideal that will never be reached.
![:diamond_shape_with_a_dot_inside: :diamond_shape_with_a_dot_inside:]() What is actual yield and theoretical yield?
What is actual yield and theoretical yield?
Determination of the real return. Actual yield is the amount of product obtained from a chemical reaction. In contrast, the calculated or theoretical yield is the amount of product that could be obtained from the reaction if all the reactants were converted to product. Theoretical performance is based on the limiting reagent.