H2s Ionic Or Covalent - How To Discuss
H2s Ionic Or Covalent
What kind of bond does hydrogen sulfide (h2s) have?
Short Answer: Hydrogen bonding forms between two molecules when they contain hydrogen and some of the three electronegative atoms (N, O, F) bond covalently. Since there is no (NOF) in H2S, there is no hydrogen bond there, even though it has dipole-dipole forces.
So what kind of bond is there in hydrogen sulfide h2s?
When atoms have very similar electronegativities, they form non-polar covalent bonds. In H2S the S atom is bonded to 2 H atoms, the electronegativity of H = 2.2 and S = 2.56.
What kind of compound is hydrogen sulfide besides the above compounds?
Hydrogen sulfide, chemical compound, H 2 S, a colorless, extremely poisonous gas that has a very unpleasant smell, just like that of rotten eggs. It is moderately soluble in water and soluble in carbon disulfide. When dissolved in water, it forms a very weak dibasic acid, sometimes called sulfuric acid.
One might also ask whether hydrogen sulfide is ionic or covalent?
Hydrogen sulfide is a covalent hydride chemically related to water (H2O) since oxygen and sulfur belong to the same group on the periodic table.
Does H2s have a polar bond?
Hydrogen sulfide is not polar. While the molecular geometry allows for polarization, the bonds are not polar and neither are the molecule. If two atoms have an electronegativity difference greater than 0.4, the bond is polar. If the difference is 0.4 or less, the bond is not polar.
Is HCL polar or non-polar?
It’s actually just that HCL is a polar molecule because chlorine has a higher electronegativity than hydrogen. It then attracts electrons to pass more time, giving it a negative charge and hydrogen a positive charge. HCL is neither polar nor apolar.
Is HCL a hydrogen bond?
Answer and explanation:
Is Sh a polar bond?
However, the difference in polarity between H and S is very small, so neither the bond nor the molecule is very polar. The longer version: if this were due solely to the polar SH bonds, the dipole of the SH bond would be at most 0.6 D with the negative end of the S atom.
What kind of bond is HCl?
covalent bond
is BCl3 polar or non-polar?
Is CBr4 polar or non-polar?
In CBr4, all bonds are polar and identical (CBr). The bonds are symmetrically arranged around the central atom of C and since the bond dipoles are disturbed, the molecule is not polar.
Is CO2 polar or non-polar?
Carbon dioxide is non-polar due to the symmetry of the bond. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are 180 degrees apart, so the dipoles are broken.
Is po4 polar or non-polar?
Answer and explanation:
Is CO2 a covalent bond?
Note that carbon dioxide has two covalent bonds between each oxygen atom and the carbon atom, shown here as two lines and referred to as a double bond. However, when the molecules are symmetrical, the atoms attract electrons in the same way and the charge distribution is the same. Symmetrical molecules are not polar.
Why is ch4 not ionic?
Is H2O an ion?
H2O is a polar covalent compound due to the different electronegativity of hydrogen (less electronegative) and oxygen (more electronegative). Due to the presence of oxygen, H2O has a partially ionic character. is
ch4 an ionic bond?
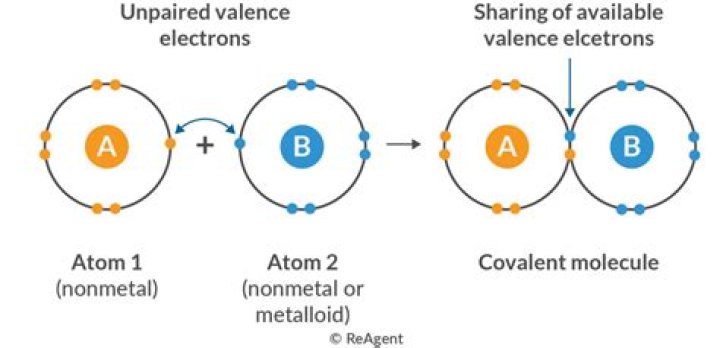
The structure of the CH4 methane molecule shows covalent bonds. Covalent bonding involves the sharing of electrons. In the methane molecule, the four hydrogen atoms share an electron with the carbon atom. Ionic bonds occur between a metal and a non-metal.
Is NaCl a covalent bond?
Ionic bonds generally occur between metallic and non-metallic ions. For example, sodium (Na), a metal, and chloride (Cl), a non-metal, form an ionic bond to form NaCl. In a covalent bond, atoms connect by sharing electrons. Covalent bonds generally occur between non-metals.
Why is water covalent and non-ionic?
Why is water covalent and non-ionic?
The electronegativity difference between oxygen and hydrogen is too small for the oxygen atom to fully extract electrons from the hydrogen atoms. NaCl, on the other hand, consists of sodium (very low electronegativity) and chlorine (very strong electronegativity).
How fast will hydrogen sulfide kill you?
What is h2s called?
So H2S is sulfuric acid instead of hydrosulfite, as the name sulfide suggests. The H2S compound is called hydrogen sulfide in its pure form, but when studying its acidic properties in aqueous solution it is called sulfuric acid.
Is H2s an acid or a base?
H2s Ionic Or Covalent
H2s Ionic Or Covalent
Is the SH bond in hydrogen sulfide non-polar, polar harmony, or ionic? 3
I'm having trouble with my high school biology homework.
Hydrogen sulfide is H2S.
The bond between SH consists of common electrons. So a harmonious bond.
Sulr is now more electronegative than hydrogen, which means it attracts electrons more strongly than hydrogen. Therefore, Hydrogen has a slightly positive charge and Sulr has a slightly negative charge.
After covalent bonding, Sulr now has 2 bound pairs and 4 unbonded electrons, which also means that 4 electrons become 2 regions of electronegativity. So Sulr has a total of 4 electron density regions. This means that the alignment of the lines forms a triangular pyramid. Since the 2 regions are pairs of electrons, the shape of the molecule is curved.
As the molecule is tilted, the polar moment of the HS bond is not lost, so H2S is polar.
Hydrogen sulfide is polar alignment.
No, ion bonds combine ions with larger charges in larger structures. Things that bind with electrons move ionically, elements that are bond-sharing electrons in harmony. The ionic bond structure is held together by large electrostatic forces and in the form of a network. Lewis's Structure: The ionic bond dot diagram shows two separate elements with electrons, one dot, the other intersects and the transferred electrons appear in the other element, because it is a dot and the other in the electron ss. - Power will be disconnected. Shows overlay overlapping elements and common electron coordinate points and cross diagrams.
The two share electrons and, because they are harmoniously bound, they are polar because sulfur has more electrons than hydrogen! Therefore, hydrogen has a slightly positive charge and sulfur has a slightly negative charge. This is because sulfur attracts more electrons than hydrogen, so polarity!
I think apolar and covalent

