Brf4 Lewis Structure - How To Discuss
By Isabella Turner •
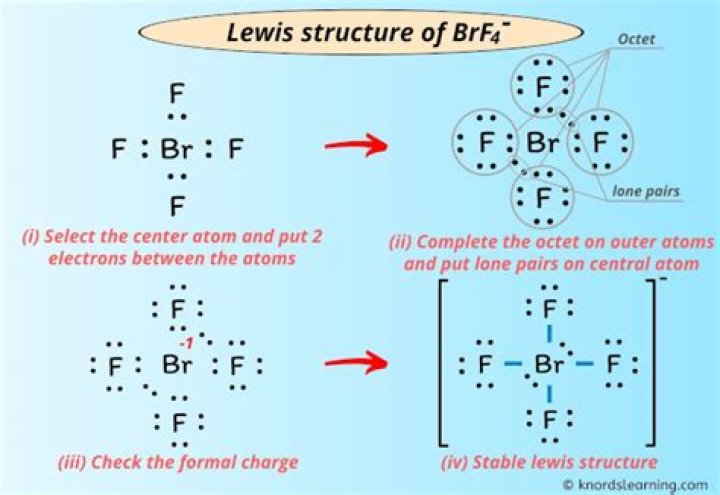
Brf4 Lewis Structure
Did you draw the Lewis structure of BrF4 +? 3
updateStretch the Lewis structure (including all single pairs) for the BrF4 + species. Keep the global charge on the central atom.
the right way?
Okay ... I'm pretty sure Br is in the middle. Draw a single bond on each F (circle each F with 3 single pairs) and leave single pairs on Br. Br +1 is charged.
Lewis framework for Brf4
Lewis's structure. Brf4
This page can help you.
D:
Did you draw the Lewis structure of BrF4 +?
Keep Br in the middle as it is the most electric. Count the valence electrons. 7 + 7 + 7 + 7 + 71 = 34. Make a bond between each Br and F (4 bonds) and add 6 electrons to each F. That means 32. Insert the last 2 valence electrons into Br a +. Last image in the upper right corner.
Brf4 Lewis Structure
Brf4 Lewis Structure
BrF4